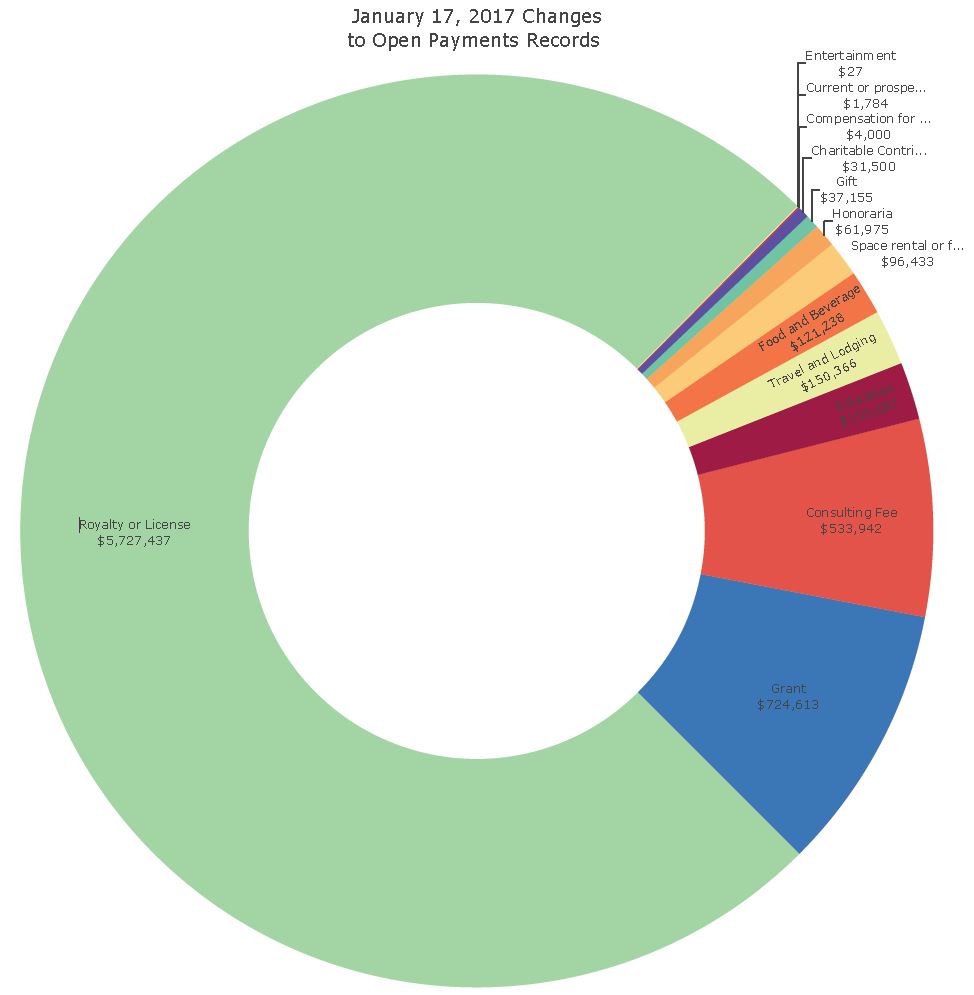
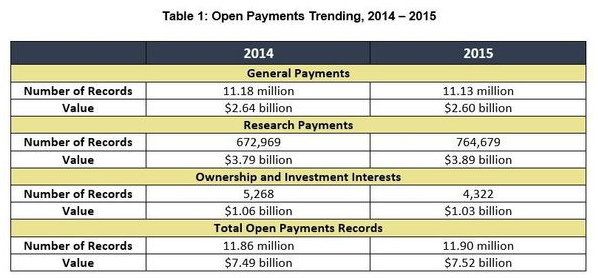
The U.S. Centers for Medicare and Medicaid Services (CMS) released Open Payments data reflecting payments and transfers of value from drug and medical device companies to physicians and teaching hospitals in 2018, totaling approximately $9.35 billion in payments and more than 11.4 million payment records. This represents a 4.3% increase from the $8.4 billion reported in 2017.