In March 2016, the Austrian Supreme Court granted an award for pain and suffering to a patient for mental strain after a piece of broken scissors was left in his body following a surgery despite the fact that the claimant had not suffered any physical pain.
reimbursement and payment
HHS announces near-doubling of civil penalties for numerous infractions
Last week, HHS, OIG, CMS, the Office of the Assistant Secretary for Financial Resources, and the Administration for Children and Families issued an interim final rule (the Rule) that adjusts maximum civil monetary penalties for inflation.
FCA case involving interpretation of 60 day overpayment rule settles
Last week, the United States and New York announced a settlement with Mount Sinai Health System for $3 million dollars to resolve allegations that Mount Sinai violated the False Claims Act by retaining overpayments for longer than 60 days after identifying them. The settlement is the first of its kind regarding the Affordable Care Act’s creation of FCA liability for providers that have identified overpayments but have not refunded such payments within 60 days.
Administrative dispute resolution process is proposed to resolve 340B drug pricing program disputes
On August 11 the Health Resources and Services Administration (HRSA) issued a notice of proposed rulemaking (RIN 0906-AA90) that would implement an administrative dispute resolution (ADR) process that would apply to all drug manufacturers and covered entities that participate in…
CMS investment in preventing healthcare fraud brings $42 billion return
CMS’s annual report demonstrates that the government is becoming increasingly effective at ferreting out fraud and abuse from the healthcare system. With a 12-to-1 return on investment, the government will likely continue to make fighting healthcare fraud a priority.
Texas supreme court temporarily halts state Medicaid cuts for home health therapy programs for disabled children


The Texas Supreme Court has temporarily blocked the Texas Health and Human Services Commission’s planned rate reductions for Medicaid home health services for severely disabled children, holding that a temporary injunction issued by a trial court enjoining the rate cuts from taking effect remains binding pending the issuance of a mandate by the Texas Third Court of Appeals reversing the trial court decision or further review by the Texas Supreme Court.
CY 2017 OPPS Proposed Rule sets new off-campus HOPD requirements

On July 6, 2016, CMS published its Calendar Year (CY) 2017 Outpatient Prospective Payment System (OPPS) Proposed Rule. We provide a more detailed analysis of the significant proposals to our July 6 post below.
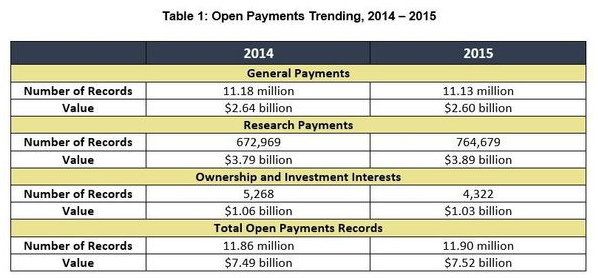
2015 CMS “Open Payments” data released
Last week, CMS posted the 2015 Open Payments data to its website, as well as updated payment records for 2013 and 2014. Overall, manufacturers reported $7.52 billion in payments and ownership interests to 618,931 physicians and 1,116 teaching hospitals in 2015. Nearly 1,500 companies reported making payments in 2015, comprising a total 11.9 million records. Although the total number of records and amounts of payment are comparable from 2014 to 2015, there are shifts in the reporting of the nature of payments.
OIG finds ED drug-discount program would not trigger kickback sanctions
On June 27, 2016, the OIG posted Advisory Opinion No. 16-07 stating that an erectile dysfunction (ED) drug manufacturer would not face sanctions for a planned discount-card promotion for the drug.
CMS publishes final rule to update medicare clinical diagnostic laboratory tests payment system for program savings

On June 17, 2016, CMS published the “Medicare Clinical Diagnostic Laboratory Tests Payment System” final rule. Among the key changes in this final rule, CMS has revised the Medicare Clinical Laboratory Fee Schedule (CLFS) such that payment for clinical diagnostic laboratory tests (CDLTs) beginning January 1, 2018 will reflect the weighted median of private payor rates.
